Current issue
Online first
Archive
About the Journal
Editorial Office
Editorial Board
Copy right and self-archiving policy
Peer review process
Instructions for Reviewers
Printed version subscription
Abstracting and indexing
Contact
Instructions for Authors
Policies
General information
Open Access, Licensing terms, Commercial use and Copyright terms policies
Self-archiving policy and Archive policies
Article Correction and Withdrawal policy
Manuscript Submission policy
Authorship policy
Conflict of Interest policy
Language considerations policy
Plagiarism and Duplicate publications policy
Ethics policy
Review process policy
Acceptance of manuscripts policy
Online First Articles and Special Issues policies
Generative artificial intelligence (AI) policy
Advertising policy
Article publication charges
Policies
General information
Open Access, Licensing terms, Commercial use and Copyright terms policies
Self-archiving policy and Archive policies
Article Correction and Withdrawal policy
Manuscript Submission policy
Authorship policy
Conflict of Interest policy
Language considerations policy
Plagiarism and Duplicate publications policy
Ethics policy
Review process policy
Acceptance of manuscripts policy
Online First Articles and Special Issues policies
Generative artificial intelligence (AI) policy
Advertising policy
ORIGINAL PAPER
Brain-derived neurotrophic factor (BDNF) affects somatotrophic
axis activity in sheep
1
The Kielanowski Institute of Animal Physiology and Nutrition, Polish Academy of Sciences, Instytucka 3, 05-110 Jabłonna, Poland
Publication date: 2021-12-13
Corresponding author
A. Wójcik-Gładysz
The Kielanowski Institute of Animal Physiology and Nutrition, Polish Academy of Sciences, Instytucka 3, 05-110 Jabłonna, Poland
The Kielanowski Institute of Animal Physiology and Nutrition, Polish Academy of Sciences, Instytucka 3, 05-110 Jabłonna, Poland
J. Anim. Feed Sci. 2021;30(4):329-339
KEYWORDS
TOPICS
ABSTRACT
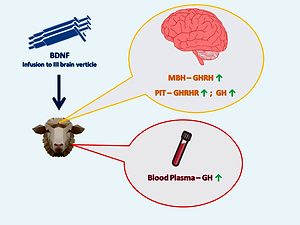
Brain-derived neurotrophic factor (BDNF) is a growth protein factor
belonging to the neurotrophin peptide family that is involved in the regulation
of several activities of the central nervous system. However, its effects on the
activity of the somatotrophic axis in ruminants have not yet been determined.
The present study aimed to verify the research hypothesis, which assumed
that BDNF can modulate key hormones of the somatotrophic axis in sheep.
The experiment involved sexually mature Polish Merino sheep (n = 24). The
animals were divided into three groups, and the following intracerebroventricular
infusions were performed: control group received Ringer-Locke solution (480
μl/day), BDNF10 – BDNF at a dose of 10 μg/480 μl/day and BDNF60 – BDNF
at a dose of 60 μg/480 μl/day. Blood samples were collected on days 0 and 3
of infusions. After the experiment, the animals were slaughtered and selected
structures of the hypothalamus, pituitaries and plasma samples were stored
for Real-Time reverse transcription-polymerase chain reaction (Real-Time
RT-PCR) and radioimmunoassay analysis. It was revealed that central BDNF
administration induced a dose-dependent increase in growth hormone relasing
hormone (GHRH) mRNA expression in the mediobasal hypothalamus of sheep.
Moreover, central BDNF administration increased growth hormone (GH) mRNA
expression in the pituitary and the GH pulse amplitude, which, consequently,
increased the mean GH concentration in the blood plasma. These observations
suggest the up-regulation of the somatotrophic axis activity in female sheep after
BDNF infusion. So, it can be stated that BDNF is involved in the modulation of
key hormones co-creating the somatotrophic axis in mature sheep.
ACKNOWLEDGEMENTS
The authors would like to thank veterinary surgeons K. Roszkowicz-Ostrowska and J. Rutkowski for the help in brain surgery, E. Skrzeczyńska, A. Misztal and K. Biernacka for the help in performing the experiments, and W. Mrozek and R. Druchniak for animal care. Presented study is the part of B.J. Przybył PhD dissertation.
FUNDING
This research was supported by the funds provided by the National Science Centre (Poland) PRELUDIUM 9 grant no. 2015/17/N/NZ9/01110.
CONFLICT OF INTEREST
The authors declared that there is no conflict of interest.
REFERENCES (48)
1.
An J.J., Liao G.Y., Kinney C.E., Sahibzada N., Xu B., 2015. Discrete BDNF neurons in the paraventricular hypothalamus control feeding and energy expenditure. Cell Metab. 22, 175–188, https://doi.org/10.1016/j.cmet....
2.
Barde Y.A., Edgar D., Thoenen H., 1982. Purification of a new neurotrophic factor from mammalian brain. EMBO J. 1, 549–553, https://doi.org/10.1002/j.1460....
3.
Barnea A., Roberts J., 2001. Induction of functional and morphological expression of neuropeptide Y (NPY) in cortical cultures by brain-derived neurotrophic factor (BDNF): evidence for a requirement for extracellular-regulated kinase (ERK)-dependent and ERK-independent mechanisms. Brain Res. 919, 57–69, https://doi.org/10.1016/S0006-....
4.
Byerly M.S., Simon J., Lebihan-Duval E., Duclos M.J., Cogburn L.A., Porter T.E., 2009. Effects of BDNF, T3, and corticosterone on expression of the hypothalamic obesity gene network in vivo and in vitro. Am. J. Physiol. Regul. Integr. Comp. Physiol. 296, R1180–R1189, https://doi.org/10.1152/ajpreg....
5.
Cordeira J.W., Frank L., Sena-Esteves M., Pothos E.N., Rios M., 2010. Brain-derived neurotrophic factor regulates hedonic feeding by acting on the mesolimbic dopamine system. J. Neurosci. 30, 2533–2541, https://doi.org/10.1523/JNEURO....
6.
Givalois L., Naert G., Tapia-Arancibia L., Arancibia S., 2006. Involvement of brain-derived neurotrophic factor in the regulation of hypothalamic somatostatin in vivo. J. Endocrinol. 188, 425–433, https://doi.org/10.1677/joe.1.....
7.
Gładysz A., Krejči P., Šimůnek J., Polkowska J., 2001. Effects of central infusions of neuropeptide Y on the somatotropic axis in sheep fed on two levels of protein. Acta Neurobiol. Exp. (Wars.) 61, 255–266.
8.
Goodman R.L., Karsch F.J., 1980. Pulsatile secretion of luteinizing hormone: differential suppression by ovarian steroids. Endocrinology 107, 1286–1290, https://doi.org/10.1210/endo-1....
9.
Hassouna R., Zizzari P., Viltart O., Yang S.K., Gardette R., Videau C., Badoer E., Epelbaum J., Tolle V., 2012. A natural variant of obestatin, Q90L, inhibits ghrelin’s action on food intake and GH secretion and targets NPY and GHRH neurons in mice. PLOS ONE 7 (12) e51135, https://doi.org/10.1371/journa....
10.
Herman A.P., Wojtulewicz K., Bochenek J., Krawczyńska A., Antushevich H., Pawlina B., Zielińska-Górska M., Herman A., Romanowicz K., Tomaszewska-Zaremba D., 2017. Endotoxin-induced inflammation disturbs melatonin secretion in ewe. Asian-Australas. J. Anim. Sci. 30, 1784–1795, https://doi.org/10.5713/ajas.1....
11.
Kennedy D., 2012. Sheep Reproduction Basics and Conception Rates. Ministry of Agriculture, Food and Rural Affairs. Guelph, Ontario (Canada), http://www.omafra.gov.on.ca/en....
12.
Krawczyńska A., Antushevich H., Bochenek J., Wojtulewicz K., Pawlina B., Herman A.P., Zięba D.A., 2019. Photoperiodic conditions as a factor modulating leptin influence on pro-inflammatory cytokines and their receptors gene expression in ewe’s aorta. J. Anim. Feed Sci. 28, 128–137, https://doi.org/10.22358/jafs/....
13.
Liu Y., Wang C., Guo G., Huo W., Zhang Y., Pei C., Liu Q., Zhang S., 2019. Growth performance of post-weaned Holstein male calves accelerated by branched-chain volatile fatty acids addition with up-regulated hepatic mTOR expression via insulin and insulin-like growth factor-1 (IGF-1) signalling pathway. J. Anim. Feed Sci. 28, 244–253, https://doi.org/10.22358/jafs/....
14.
Luque R.M., Park S., Kineman R.D., 2008. Role of endogenous somatostatin in regulating GH output under basal conditions and in response to metabolic extremes. Mol. Cell Endocrinol. 286, 155–168, https://doi.org/10.1016/j.mce.....
15.
Marmigère F., Rage F., Tapia-Arancibia L., 2001. Regulation of brain-derived neurotrophic factor transcripts by neuronal activation in rat hypothalamic neurons. J. Neurosci. Res. 66, 377–389, https://doi.org/10.1002/jnr.12....
16.
Nawa H., Pelleymounter M., Carnahan J., 1994. Intraventricular administration of BDNF increases neuropeptide expression in newborn rat brain. J. Neurosci. 14, 3751–3765, https://doi.org/10.1523/JNEURO....
17.
Nicholson J.R., Peter J.C., Lecourt A.C., Barde Y.A., Hofbauer K.G., 2007. Melanocortin-4 receptor activation stimulates hypothalamic brain-derived neurotrophic factor release to regulate food intake, body temperature and cardiovascular function. J. Neuroendocrinol. 19, 974–982, https://doi.org/10.1111/j.1365....
18.
Nogueiras R., Pfluger P., Tovar S. et al., 2007. Effects of obestatin on energy balance and growth hormone secretion in rodents. Endocrinology 148, 21–26, https://doi.org/10.1210/en.200....
19.
Patapoutian A., Reichardt L.F., 2001. Trk receptors: mediators of neurotrophin action. Curr. Opin. Neurobiol. 11, 272–280, https://doi.org/10.1016/S0959-....
20.
Peiris T.S., Machaalani R., Waters K.A., 2004. Brain-derived neurotrophic factor mRNA and protein in the piglet brainstem and effects of Intermittent Hypercapnic Hypoxia. Brain Res. 1029, 11–23, https://doi.org/10.1016/j.brai....
21.
Pfaffl M.W., Horgan G.W., Dempfle L., 2002. Relative expression software tool (REST©) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res. 30, e36, https://doi.org/10.1093/nar/30....
22.
Pfaffl M.W., Tichopad A., Prgomet C., Neuvians T.P., 2004. Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper – Excel-based tool using pair-wise correlations. Biotechnol. Lett. 26, 509–515, https://doi.org/10.1023/b:bile....
23.
Polkowska J., Wańkowska M., 2010. Effects of maternal deprivation on the somatotrophic axis and neuropeptide Y in the hypothalamus and pituitary in female lambs. The histomorphometric study. Folia Histochem. Cytobiol. 48, 299–305, https://doi.org/10.2478/v10042....
24.
Polkowska J., Wójcik-Gładysz A., Wańkowska M., Bruneau G., Tillet Y., 2008. Prepubertal changes in the synthesis, storage and release of the somatostatin in the hypothalamus of female lambs: A morphofunctional study. J. Chem. Neuroanat. 36, 53–58 https://doi.org/10.1016/j.jche....
25.
Przybył B.J., Szlis M., Wójcik-Gładysz A., 2020. Brain-derived neurotrophic factor affects mRNA and miRNA expression of the appetite regulating centre in the sheep arcuate nucleus. Ann. Anim. Sci. 20, 853–869, https://doi.org/10.2478/aoas-2....
26.
Przybył B.J., Szlis M., Wójcik-Gładysz A., 2021. Brain-derived neurotrophic factor (BDNF) affects the activity of the gonadotrophic axis in sheep. Horm. Behav. 131, 104980, https://doi.org/10.1016/j.yhbe....
27.
Rekešiutė A., Januškauskas A., Kerzienė S., Sutkevieìenė N., Vaieiùlienė G., Rutkauskas A., Žilinskas H., 2020. Evaluation of efficacy of selected estrous synchronization programs depending on the functional state of the ovaries, heifers and cows of meat breed. J. Anim. Feed Sci. 29, 175–179, https://doi.org/10.22358/jafs/....
28.
Sibilia V., Brescian E., Lattuada N., Rapetti D., Locatelli V., De Luca V., Donà C., Netti A., Torsello F., Guidobono F., 2006. Intracerebroventricular acute and chronic administration of obestatin minimally affect food intake but not weight gain in the rat. J. Endocrinol. Invest. 29, RC31–RC34, https://doi.org/10.1007/BF0334....
29.
Slaba J., Krejcí P., Skarda J., Huybrechts L.M., Decuypere E., Herrmann H., 1994. Plasma profiles of somatotropin and IGF-I in diary cows following application of two preparations of recombinant bovine somatotropin in a sustained release vehicle. Physiol. Res. 43, 37–43.
30.
Strzetelski J.A., Brzóska F., Kowalski Z.M., Osięgłowski S., 2014. Feeding Recommendation for Ruminants and Feed Tables (in Polish: Zalecenia Żywieniowe dla Przeżuwaczy i Tabele wartości pokarmowej pasz). National Research Institute of Animal Production. Krakow (Poland).
31.
Szlis M., Polkowska J., Skrzeczyńska E., Przybył B.J., Wójcik-Gładysz A., 2018. Does obestatin modulate the hypothalamic appetite-regulating network in peripubertal sheep? J. Anim. Physiol. Anim. Nutr. 102, 690–700, https://doi.org/10.1111/jpn.12....
32.
Szlis M., Wójcik-Gładysz A., Przybył B.J., 2020. Central obestatin administration affect the LH and FSH secretory activity in peripubertal sheep. Theriogenology 145, 10–17, https://doi.org/10.1016/j.ther....
33.
Takei N., Furukawa K., Hanyu O., Sone H., Nawa H., 2014. A possible link between BDNF and mTOR in control of food intake. Front. Psychol. 5, 1093, https://doi.org/10.3389/fpsyg.....
34.
Tillet Y., Picard S., Bruneau G., Ciofi P., Wańkowska M., Wójcik-Gładysz A., Polkowska J., 2010. Hypothalamic arcuate neuropeptide Y-neurons decrease periventricular somatostatin-neuronal activity before puberty in the female lamb: Morphological arguments. J. Chem. Neuroanat. 40, 265–271 https://doi.org/10.1016/j.jche....
35.
Timmusk T., Palm K., Metsis M., Reintam T., Paalme V., Saarma M., Persson H., 1993. Multiple promoters direct tissue-specific expression of the rat BDNF gene. Neuron 10, 475–489, https://doi.org/10.1016/0896-6....
36.
Traczyk W., Przekop F., 1963. Methods of investigation of the function of the hypothalamus and hypophysis in chronic experiments in sheep (in Polish). Acta. Physiol. Pol. 14, 217–226.
37.
Unger T.J., Calderon G.A., Bradley L.C., Sena-Esteves M., Rios M., 2007. Selective deletion of Bdnf in the ventromedial and dorsomedial hypothalamus of adult mice results in hyperphagic behavior and obesity. J. Neurosci. 27, 14265–14274, https://doi.org/10.1523/JNEURO....
38.
Welento J., Szteyn S., Milart Z., 1969. Observations on the stereotaxic configuration of the hypothalamus nuclei in the sheep. Anat. Anz. 124, 1–27.
39.
Wójcik-Gładysz A., Szlis M., 2016. Hypothalamo-gastrointestinal axis – role in food intake regulation. J. Anim. Feed Sci. 25, 97–108, https://doi.org/10.22358/jafs/....
40.
Wójcik-Gładysz A., Szlis M., Misztal A., Przybył B.J., Polkowska J., 2018. Obestatin stimulates the somatotrophic axis activity in sheep. Brain Res. 1678, 278–287, https://doi.org/10.1016/j.brai....
41.
Wójcik-Gładysz A., Szlis M., Przybył B.J., Polkowska J., 2019. Obestatin may affect the GnRH/KNDy gene network in sheep hypothalamus. Res. Vet. Sci. 123, 51–58, https://doi.org/10.1016/j.rvsc....
42.
Wójcik-Gładysz A., Wańkowska M., Gajewska A., Misztal T., Szlis M., Polkowska J., 2014. The effect of intracerebroventricular infusions of ghrelin on the secretory activity of the GnRH/LH system in peripubertal ewes. J. Anim. Feed Sci. 23, 299–308, https://doi.org/10.22358/jafs/....
43.
Wójcik-Gładysz A., Wańkowska M., Gajewska A., Misztal T., Zielińska-Górska M., Szlis M., Polkowska J., 2016. Effects of intracerebroventricular infusions of ghrelin on secretion of follicle-stimulating hormone in peripubertal female sheep. Reprod. Fertil. Dev. 28, 2065–2074, https://doi.org/10.1071/RD1602....
44.
Wójcik-Gładysz A., Wańkowska M., Misztal T., Szczepankiewicz D., Romanowicz K., Polkowska J., 2010. The effect of intracerebroventricular infusion of leptin on the secretory activity of the somtotropic axis in fasted prepubertal lambs. J. Anim. Feed Sci. 19, 379–397, https://doi.org/10.22358/jafs/....
45.
Xapelli S., Bernardino L., Ferreira R. et al., 2008. Interaction between neuropeptide Y (NPY) and brain-derived neurotrophic factor in NPY-mediated neuroprotection against excitotoxicity: a role for microglia. Eur. J. Neurosci. 27, 2089–2102, https://doi.org/10.1111/j.1460....
46.
Yamamoto D., Ikeshita N., Daito R., et al., 2007. Neither intravenous nor intracerebroventricular administration of obestatin affects the secretion of GH, PRL, TSH and ACTH in rats. Regul. Pept. 138, 141–144, https://doi.org/10.1016/j.regp....
47.
Yoshihara Y., Ogawa Y., 2021. Handling stress-induced rumianal microbiota changes reduce grass hay degradability in sheep. J. Anim. Feed Sci. 30, 159–164, https://doi.org/10.22358/jafs/....
48.
Zizzari P., Longchamps R., Epelbaum J., Bluet-Pajot M.T., 2007. Obestatin partially affects ghrelin stimulation of food intake and growth hormone secretion in rodents. Endocrinology 148, 1648–1653, https://doi.org/10.1210/en.200....
CITATIONS (12):
1.
The Effect of Leptin on the Blood Hormonal Profile (Cortisol, Insulin, Thyroid Hormones) of the Ewe in Acute Inflammation in Two Different Photoperiodical Conditions
Agata Krawczyńska, Andrzej Herman, Hanna Antushevich, Joanna Bochenek, Karolina Wojtulewicz, Dorota Zieba
International Journal of Molecular Sciences
Agata Krawczyńska, Andrzej Herman, Hanna Antushevich, Joanna Bochenek, Karolina Wojtulewicz, Dorota Zieba
International Journal of Molecular Sciences
2.
Therapeutic effects of growth hormone in a rat model of total sleep deprivation: Evaluating behavioral, hormonal, biochemical, electrophysiological and oxidative stress markers
Parisa Arvin, Samireh Ghafouri, Kosar Bavarsad, Somayeh Hajipour, Seyed Khoshnam, Alireza Sarkaki, Yaghoub Farbood
Behavioural Brain Research
Parisa Arvin, Samireh Ghafouri, Kosar Bavarsad, Somayeh Hajipour, Seyed Khoshnam, Alireza Sarkaki, Yaghoub Farbood
Behavioural Brain Research
3.
Central Stimulatory Effect of Kynurenic Acid on BDNF-TrkB Signaling and BER Enzymatic Activity in the Hippocampal CA1 Field in Sheep
Katarzyna Roszkowicz-Ostrowska, Patrycja Młotkowska, Paweł Kowalczyk, Elżbieta Marciniak, Marcin Barszcz, Tomasz Misztal
International Journal of Molecular Sciences
Katarzyna Roszkowicz-Ostrowska, Patrycja Młotkowska, Paweł Kowalczyk, Elżbieta Marciniak, Marcin Barszcz, Tomasz Misztal
International Journal of Molecular Sciences
4.
Influence of Leptin on the Secretion of Growth Hormone in Ewes under Different Photoperiodic Conditions
Maciej Wójcik, Agata Krawczyńska, Dorota Zieba, Hanna Antushevich, Andrzej Herman
International Journal of Molecular Sciences
Maciej Wójcik, Agata Krawczyńska, Dorota Zieba, Hanna Antushevich, Andrzej Herman
International Journal of Molecular Sciences
5.
Circadian and seasonal changes
in the expression of clock genes in the ovine pars tuberalis
K. Wojtulewicz, M. Tomczyk, M. Wójcik, J. Bochenek, H. Antushevich, A. Krawczyńska, M. Załęcki, A. Herman
Journal of Animal and Feed Sciences
K. Wojtulewicz, M. Tomczyk, M. Wójcik, J. Bochenek, H. Antushevich, A. Krawczyńska, M. Załęcki, A. Herman
Journal of Animal and Feed Sciences
6.
Time-dependent effect of inflammation on the gene expression
of pro-inflammatory cytokines and their receptors at the different
levels of the somatotropic axis in ewe
M. Wójcik, D. Zięba, M. Tomczyk, J. Bochenek, H. Antushevich, A. Krawczyńska, A. Herman
Journal of Animal and Feed Sciences
M. Wójcik, D. Zięba, M. Tomczyk, J. Bochenek, H. Antushevich, A. Krawczyńska, A. Herman
Journal of Animal and Feed Sciences
7.
The role of QRFP43 in the secretory activity of the gonadotrophic axis in female sheep
Bartosz Jarosław Przybył, Michał Szlis, Bartłomiej Wysoczański, Anna Wójcik-Gładysz
Scientific Reports
Bartosz Jarosław Przybył, Michał Szlis, Bartłomiej Wysoczański, Anna Wójcik-Gładysz
Scientific Reports
8.
The Effect of Endotoxin‐Induced Inflammation on the Activity of the Somatotropic Axis in Sheep
Maciej Wójcik, Dorota Anna Zieba, Joanna Bochenek, Agata Krawczyńska, Marcin Barszcz, Alina Gajewska, Hanna Antushevich, Andrzej Przemysław Herman, Mariusz Sikora
International Journal of Inflammation
Maciej Wójcik, Dorota Anna Zieba, Joanna Bochenek, Agata Krawczyńska, Marcin Barszcz, Alina Gajewska, Hanna Antushevich, Andrzej Przemysław Herman, Mariusz Sikora
International Journal of Inflammation
9.
Organ-On-A-Chip Models—New Possibilities in Experimental Science and Disease Modeling
Bartłomiej Wysoczański, Marcin Świątek, Anna Wójcik-Gładysz
Biomolecules
Bartłomiej Wysoczański, Marcin Świątek, Anna Wójcik-Gładysz
Biomolecules
10.
Dietary Patterns, Serum BDNF and Fatty Acid Profiles in Physically Active Male Young Adults: A Cluster Analysis Study
Monika Johne, Ewelina Maculewicz, Andrzej Mastalerz, Małgorzata Białek, Wiktoria Wojtak, Bartosz Osuch, Małgorzata Majewska, Marian Czauderna, Agnieszka Białek
Nutrients
Monika Johne, Ewelina Maculewicz, Andrzej Mastalerz, Małgorzata Białek, Wiktoria Wojtak, Bartosz Osuch, Małgorzata Majewska, Marian Czauderna, Agnieszka Białek
Nutrients
11.
QRFP43 modulates somatotrophic axis activity in female sheep
M. Szlis, B. J. Przybył, A. Wójcik-Gładysz
Journal of Animal and Feed Sciences
M. Szlis, B. J. Przybył, A. Wójcik-Gładysz
Journal of Animal and Feed Sciences
12.
Effect of Central Injection of Anandamide on LPS-Dependent Suppression of GnRH/LH Secretion in Ewes During the Follicular Phase of the Estrous Cycle
Karolina Wojtulewicz, Dorota Tomaszewska-Zaremba, Monika Tomczyk, Joanna Bochenek, Andrzej P. Herman
International Journal of Molecular Sciences
Karolina Wojtulewicz, Dorota Tomaszewska-Zaremba, Monika Tomczyk, Joanna Bochenek, Andrzej P. Herman
International Journal of Molecular Sciences
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.