Current issue
Online first
Archive
About the Journal
Editorial Office
Editorial Board
Copy right and self-archiving policy
Peer review process
Instructions for Reviewers
Printed version subscription
Abstracting and indexing
Contact
Instructions for Authors
Policies
General information
Open Access, Licensing terms, Commercial use and Copyright terms policies
Self-archiving policy and Archive policies
Article Correction and Withdrawal policy
Manuscript Submission policy
Authorship policy
Conflict of Interest policy
Language considerations policy
Plagiarism and Duplicate publications policy
Ethics policy
Review process policy
Acceptance of manuscripts policy
Online First Articles and Special Issues policies
Generative artificial intelligence (AI) policy
Advertising policy
Article publication charges
Policies
General information
Open Access, Licensing terms, Commercial use and Copyright terms policies
Self-archiving policy and Archive policies
Article Correction and Withdrawal policy
Manuscript Submission policy
Authorship policy
Conflict of Interest policy
Language considerations policy
Plagiarism and Duplicate publications policy
Ethics policy
Review process policy
Acceptance of manuscripts policy
Online First Articles and Special Issues policies
Generative artificial intelligence (AI) policy
Advertising policy
ORIGINAL PAPER
Effect of caffeine on adenosine and ryanodine receptor
gene expression in the hypothalamus, pituitary, and choroid
plexus in ewes under basal and LPS challenge conditions
1
Institute of Animal Reproduction and Food Research, Polish Academy of Sciences, 10-748 Olsztyn, Poland
2
The Kielanowski Institute of Animal Physiology and Nutrition, Polish Academy of Sciences, 05-110 Jabłonna, Poland
Publication date: 2022-12-20
Corresponding author
A. P. Herman
The Kielanowski Institue of Animal Physiology and Nutrition, Polish Academy of Sciences
The Kielanowski Institue of Animal Physiology and Nutrition, Polish Academy of Sciences
J. Anim. Feed Sci. 2023;32(1):17-25
KEYWORDS
TOPICS
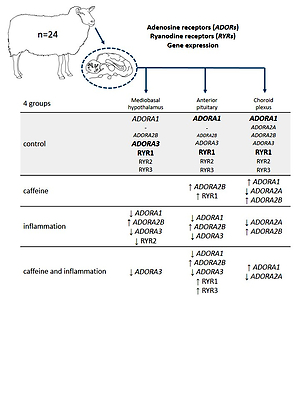
ABSTRACT
Caffeine is a plant alkaloid that stimulates the central nervous
system. It easily crosses the blood-brain barrier located in the endothelial cells
of brain microvessels and the blood-cerebrospinal fluid barrier located in the
epithelial cells of the choroid plexus (ChP). Caffeine exerts most of its biological
effects by antagonising adenosine receptors (ADORs), but is also an agonist of
ryanodine receptors (RYRs). A recent study in a sheep model has suggested
that the effect of caffeine on the expression of many genes may depend on the
animal’s immune status. Therefore, the aim of the study was to determine the
effect of caffeine administration (iv, 30 mg/kg) in ewes, under basal and acute
inflammatory conditions induced by lipopolysaccharide injection (iv, 400 ng/kg),
on the expression of ADOR and RYR genes in the mediobasal hypothalamus
(MBH), anterior pituitary (AP), and ChP. Our study showed that among caffeineinteracting
receptors, ADORA1 was the most highly expressed in the AP and
ChP, while ADORA3 in the MBH. Caffeine reduced (P < 0.05) the inhibitory
effect of inflammation on ADORA1, but only in the MBH, and decreased
(P < 0.05) the stimulatory effect of endotoxin treatment on ADORA2B in the MBH
and ChP. In contrast to ADORs, the expression of RYRs was less sensitive to
the effects of inflammation and caffeine. We showed that caffeine influenced the
expression of its receptor genes in the brain, but this effect seemed to be tissuedependent
and could be affected by the immune status of the animals. However,
the physiological implications of these results require further detailed studies.
FUNDING
This work was supported by funds granted by the
National Science Centre, Poland, based on Decision
No. DEC-2017/25/B/NZ9/00225. AS and JS were
supported by statutory research funds of the Institute
of Animal Reproduction and Food Research.
CONFLICT OF INTEREST
The Authors declare that there is no conflict of
interest.
REFERENCES (35)
1.
Abu-Omar N., Das J., Szeto V., Feng Z.-P., 2018. Neuronal ryanodine receptors in development and aging. Mol. Neurobiol. 55, 1183–1192, doi.org/10.1007/s12035-016-0375-4
2.
Buenestado A., Grassin Delyle S., Arnould I., Besnard F., Naline E., Blouquit-Laye S., Chapelier A., Bellamy J.F., Devillier P., 2010. The role of adenosine receptors in regulating production of tumour necrosis factor-alpha and chemokines by human lung macrophages. Br. J. Pharmacol. 159, 1304–1311, doi.org/10.1111/j.1476-5381.2009.00614.x
3.
Chen J.F., Lee C.F., Chern Y., 2014. Chapter One – adenosine receptor neurobiology: overview. Int. Rev. Neurobiol. 119, 1–49, doi.org/10.1016/B978-0-12-801022-8.00001-5
4.
Cohen S., Fishman P., 2019. Targeting the A3 adenosine receptor to treat cytokine release syndrome in cancer immunotherapy. Drug Des. Devel. Ther. 13, 491–497, doi.org/10.2147/DDDT.S195294
5.
Cronstein B.N., Sitkovsky M., 2017. Adenosine and adenosine receptors in the pathogenesis and treatment of rheumatic diseases. Nat. Rev. Rheumatol. 13, 41–51, doi.org/10.1038/nrrheum.2016.178
6.
Endesfelder S., Strauß E., Bendix I., Schmitz T., Bührer C., 2020. Prevention of oxygen-induced inflammatory lung injury by caffeine in neonatal rats. Oxid. Med. Cell. Longev. 2020, 3840124, doi.org/10.1155/2020/3840124
7.
Fredholm B.B., Ijzerman A.P., Jacobson K.A., Linden J., Muller C.E., 2011. International Union of Basic and Clinical Pharmacology. LXXXI. Nomenclature and classification of adenosine receptors –an update. Pharmacol. Rev. 63, 1–34, doi.org/10.1124/pr.110.003285
8.
Gamble K.L, Ciarleglio C.M., 2009. Ryanodine receptors are regulated by the circadian clock and implicated in gating photic entrainment. J. Neurosci. 29, 11717–11719, doi.org/10.1523/JNEUROSCI.3820-09.2009
9.
Hackett T.A., Guo Y., Clause A., Hackett N.J., Garbett K., Zhang P., Polley D.B., Mirnics K., 2015. Transcriptional maturation of the mouse auditory forebrain. BMC Genomics 16, 606
10.
Haziak K., Herman A.P., Tomaszewska-Zaremba D., 2014. Effects of central injection of anti-LPS antibody and blockade of TLR4 on GnRH/LH secretion during immunological stress in anestrous ewes. Mediators Inflamm. 2014, 867170, https://doi.org/10.1155/2014/8...
11.
Ikeda-Murakami K., Tani N., Ikeda T., Aoki Y., Ishikawa T., 2022. Central nervous system stimulants limit caffeine transport at the blood-cerebrospinal fluid barrier. Int. J. Mol. Sci. 23, 1862, doi.org/10.3390/ijms23031862
12.
Jagannath A., Varga N., Dallmann R. et al., 2021. Adenosine integrates light and sleep signalling for the regulation of circadian timing in mice. Nat. Commun. 12, 2113, doi.org/10.1038/s41467-021-22179-z
13.
Karmouty-Quintana H., Xia Y., Blackburn M.R., 2013. Adenosine signaling during acute and chronic disease states. J. Mol. Med. (Berl.). 91, 173–181, doi.org/10.1007/s00109-013-0997-1
14.
Kong H., Jones P.P., Koop A., Zhang L., Duff H.J., Chen S.R.W., 2008. Caffeine induces Ca2+ release by reducing the threshold for luminal Ca2+ activation of the ryanodine receptor. Biochem. J. 414, 441–452, doi.org/10.1042/BJ20080489
15.
Kowalewska M., Herman A.P., Szczepkowska A., Skipor J., 2017. The effect of melatonin from slow-release implants on basic and TLR-4-mediated gene expression of inflammatory cytokines and their receptors in the choroid plexus in ewes. Res. Vet. Sci. 113, 50–55, doi.org/10.1016/j.rvsc.2017.09.003
16.
Liao J., Zhang S., Yang S. et al., 2021. Interleukin-6-mediated-Ca2+ handling abnormalities contributes to atrial fibrillation in sterile pericarditis rats. Front. Immunol. 12, 758157, doi.org/10.3389/fimmu.2021.758157
17.
Lu M., Farnebo L.-O., Bränström R., Larsson C., 2013. Inhibition of parathyroid hormone secretion by caffeine in human parathyroid cells. J. Clin. Endocrinol. Metab. 98, E1345–E1351, https://doi.org/10.1210/jc.201...
18.
McCall A.L., Millington W.R., Wurtman R.J., 1982. Blood-brain barrier transport of caffeine: dose-related restriction of adenine transport. Life Sci. 31, 2709–2715, doi.org/10.1016/0024-3205(82)90715-9
19.
Modic M.T., Weinstein M.A., Rothner A.D., Erenberg G., Duchesneau P.M., Kaufman B., 1980. Calcification of the choroid plexus visualized by computed tomography. Radiology 135, 369–372, doi.org/10.1148/radiology.135.2.7367628
20.
Murphy P.S., Wang J., Bhagwat S.P., Munger J.C., Janssen W.J., Wright T.W., Elliott M.R., 2017. CD73 regulates anti-inflammatory signaling between apoptotic cells and endotoxin-conditioned tissue macrophages. Cell Death Differ. 24, 559–570, doi.org/10.1038/cdd.2016.159
21.
Persad L.A.B., 2011. Energy drinks and the neurophysiological impact of caffeine. Front. Neurosci. 5, 116, doi.org/10.3389/fnins.2011.00116
22.
Rasmussen R., 2001. Quantification on the LightCycler. In: S. Meuer, C. Wittwer, K.I. Nakagawara (Editors). Rapid Cycle Real-Time PCR. Springer. Berlin (Germany), pp. 21–34, https://doi.org/10.1007/978-3-...
23.
Ren T.H., Lv M.M., Am X.M., Leung W.K., Seto W.K., 2020. Activation of adenosine A3 receptor inhibits inflammatory cytokine production in colonic mucosa of patients with ulcerative colitis by down-regulating the nuclear factor-kappa B signaling. J. Dig. Dis. 21, 38–45, https://doi.org/10.1111/1751-2...
24.
Ribeiro J.A., Sebastiao A.M., 2010. Caffeine and adenosine. J. Alzheimer’s Dis. 20, S3–S15, doi.org/10.3233/JAD-2010-1379
25.
Rouillard A.D., Gundersen G.W., Fernandez N.F., Wang Z., Monteiro C.D., McDermott M.G., Ma'ayan A., 2016. The harmonizome: a collection of processed datasets gathered to serve and mine knowledge about genes and proteins. Database. Oxford (UK), pii: baw100, maayanlab.cloud/Harmonizome
26.
Schiffmann S.N., Libert F., Vassart G., Vanderhaeghen J.J., 1991. Distribution of adenosine A2 receptor mRNA in the human brain. Neurosci Lett. 130, 177–181, doi.org/10.1016/0304-3940(91)90391-6
27.
Sjöstedt E., Zhong W., Fagerberg L. et al., 2020. An atlas of the protein-coding genes in the human, pig, and mouse brain. Science 367, 6482, doi.org/10.1126/science.aay5947
28.
Stamp L.K., Hazlett J., Roberts R.L., Frampton C., Highton J., Hessian P.A., 2012. Adenosine receptor expression in rheumatoid synovium: a basis for methotrexate action. Arthritis Res. Ther. 14, R138, doi.org/10.1186/ar3871
29.
Sundaresan S., Weiss J., Bauer-Dantoin A.C., Jameson J.L., 1997. Expression of ryanodine receptors in the pituitary gland: evidence for a role in gonadotropin-releasing hormone signaling. Endocrinology 138, 2056–2065, doi.org/10.1210/endo.138.5.5153
30.
Szczepkowska A., Wójcik M., Tomaszewska-Zaremba D., Antushevich H., Krawczyńska A., Wiechetek W., Skipor J., Herman A.P., 2021. Acute effect of caffeine on the synthesis of pro-inflammatory cytokines in the hypothalamus and choroid plexus during endotoxin-induced inflammation in a female sheep model. Int. J. Mol. Sci. 22, 13237, doi.org/10.3390/ijms222413237
31.
Van Eps N., Altenbach C., Caro L.N., Latorraca N.R., Hollingsworth S.A., Dror R.O., Ernst O.P., Hubbell W.L., 2018. Gi- and Gs-coupled GPCRs show different modes of G-protein binding. Proc. Natl. Acad. Sci. U.S.A. 115, 2383–2388, doi.org/10.1073/pnas.1721896115
32.
Yang D., Zhang Y., Nguyen H.G. et al, 2006. The A2B adenosine receptor protects against inflammation and excessive vascular adhesion. J. Clin. Invest. 116, 1913–1923, doi.org/10.1172/JCI27933
33.
Yang T., Gao X., Sandberg M. et al., 2015. Abrogation of adenosine A1 receptor signalling improves metabolic regulation in mice by modulating oxidative stress and inflammatory responses. Diabetologia 58, 1610–1620, doi.org/10.1007/s00125-015-3570-3
34.
Zhao S., Fernald R.D., 2005. Comprehensive algorithm for quantitative real-time polymerase chain reaction. J. Comput. Biol. 12, 1047–1064, doi.org/10.1089/cmb.2005.12.1047
35.
Zhao Y., Li M.C., Konaté M.M., Chen L., Das B., Karlovich C., Williams P.M., Evrard Y.A., Doroshow J.H., McShane L.M., 2021. TPM, FPKM, or normalized counts? A comparative study of quantification measures for the analysis of RNA-seq data from the NCI patient-derived models repository. J. Transl. Med. 19, 269, doi.org/10.1186/s12967-021-02936-w
CITATIONS (16):
1.
Influence of Leptin on the Secretion of Growth Hormone in Ewes under Different Photoperiodic Conditions
Maciej Wójcik, Agata Krawczyńska, Dorota Zieba, Hanna Antushevich, Andrzej Herman
International Journal of Molecular Sciences
Maciej Wójcik, Agata Krawczyńska, Dorota Zieba, Hanna Antushevich, Andrzej Herman
International Journal of Molecular Sciences
2.
Circadian and seasonal changes
in the expression of clock genes in the ovine pars tuberalis
K. Wojtulewicz, M. Tomczyk, M. Wójcik, J. Bochenek, H. Antushevich, A. Krawczyńska, M. Załęcki, A. Herman
Journal of Animal and Feed Sciences
K. Wojtulewicz, M. Tomczyk, M. Wójcik, J. Bochenek, H. Antushevich, A. Krawczyńska, M. Załęcki, A. Herman
Journal of Animal and Feed Sciences
3.
Time-dependent effect of inflammation on the gene expression
of pro-inflammatory cytokines and their receptors at the different
levels of the somatotropic axis in ewe
M. Wójcik, D. Zięba, M. Tomczyk, J. Bochenek, H. Antushevich, A. Krawczyńska, A. Herman
Journal of Animal and Feed Sciences
M. Wójcik, D. Zięba, M. Tomczyk, J. Bochenek, H. Antushevich, A. Krawczyńska, A. Herman
Journal of Animal and Feed Sciences
4.
Effect of Caffeine on the Inflammatory-Dependent Changes in the GnRH/LH Secretion in a Female Sheep Model
Andrzej Przemysław Herman, Monika Tomczyk, Maciej Wójcik, Joanna Bochenek, Hanna Antushevich, Anna Herman, Wiktoria Wiechetek, Aleksandra Szczepkowska, Elżbieta Marciniak, Dorota Tomaszewska-Zaremba
International Journal of Molecular Sciences
Andrzej Przemysław Herman, Monika Tomczyk, Maciej Wójcik, Joanna Bochenek, Hanna Antushevich, Anna Herman, Wiktoria Wiechetek, Aleksandra Szczepkowska, Elżbieta Marciniak, Dorota Tomaszewska-Zaremba
International Journal of Molecular Sciences
5.
The role of QRFP43 in the secretory activity of the gonadotrophic axis in female sheep
Bartosz Jarosław Przybył, Michał Szlis, Bartłomiej Wysoczański, Anna Wójcik-Gładysz
Scientific Reports
Bartosz Jarosław Przybył, Michał Szlis, Bartłomiej Wysoczański, Anna Wójcik-Gładysz
Scientific Reports
6.
Effect of kynurenic acid on enzymatic activity of the DNA base excision repair pathway in specific areas of the sheep brain
Patrycja Młotkowska, Tomasz Misztal, Paweł Kowalczyk, Elżbieta Marciniak
Scientific Reports
Patrycja Młotkowska, Tomasz Misztal, Paweł Kowalczyk, Elżbieta Marciniak
Scientific Reports
7.
Effect of central administration of indomethacin on anandamide-induced GnRH/LH secretion in the hypothalamus of anoestrous ewes
Dorota Tomaszewska-Zaremba, Monika Tomczyk, Karolina Wojtulewicz, Joanna Bochenek, Kinga Pałatyńska, Andrzej Przemysław Herman
Journal of Veterinary Research
Dorota Tomaszewska-Zaremba, Monika Tomczyk, Karolina Wojtulewicz, Joanna Bochenek, Kinga Pałatyńska, Andrzej Przemysław Herman
Journal of Veterinary Research
8.
The Effect of Endotoxin‐Induced Inflammation on the Activity of the Somatotropic Axis in Sheep
Maciej Wójcik, Dorota Anna Zieba, Joanna Bochenek, Agata Krawczyńska, Marcin Barszcz, Alina Gajewska, Hanna Antushevich, Andrzej Przemysław Herman, Mariusz Sikora
International Journal of Inflammation
Maciej Wójcik, Dorota Anna Zieba, Joanna Bochenek, Agata Krawczyńska, Marcin Barszcz, Alina Gajewska, Hanna Antushevich, Andrzej Przemysław Herman, Mariusz Sikora
International Journal of Inflammation
9.
Influence of Acute Inflammation on the Expression of Clock Genes in the Ovine Pars Tuberalis Under Different Photoperiodic Conditions
Karolina Wojtulewicz, Monika Tomczyk, Maciej Wójcik, Hanna Antushevich, Joanna Bochenek, Andrzej Przemysław Herman
International Journal of Molecular Sciences
Karolina Wojtulewicz, Monika Tomczyk, Maciej Wójcik, Hanna Antushevich, Joanna Bochenek, Andrzej Przemysław Herman
International Journal of Molecular Sciences
10.
QRFP43 modulates the activity of the hypothalamic-pituitary-thyroid axis in female sheep
Bartosz Jarosław Przybył, Michał Szlis, Anna Misztal, Anna Wójcik-Gładysz
Scientific Reports
Bartosz Jarosław Przybył, Michał Szlis, Anna Misztal, Anna Wójcik-Gładysz
Scientific Reports
11.
Effect of co-incubation of HPAd-derived adipocytes
with enterocytes on inflammatory agents expression and bacterial
adhesion in Caco-2-derived enterocytes: a preliminary study
H. Antushevich, M. Wójcik, M. Załęcki, K. Wojtulewicz, M. Matusiewicz, J. Bochenek
Journal of Animal and Feed Sciences
H. Antushevich, M. Wójcik, M. Załęcki, K. Wojtulewicz, M. Matusiewicz, J. Bochenek
Journal of Animal and Feed Sciences
12.
ANSIEDADE E ALIMENTAÇÃO
GIOVANA MARIA SILVA SOUSA, JANICIARA BOTELHO SILVA, IRIANE DO NASCIMENTO ROSA
INTERNATIONAL JOURNAL EDUCATION AND TEACHING (PDVL) ISSN 2595-2498
GIOVANA MARIA SILVA SOUSA, JANICIARA BOTELHO SILVA, IRIANE DO NASCIMENTO ROSA
INTERNATIONAL JOURNAL EDUCATION AND TEACHING (PDVL) ISSN 2595-2498
13.
QRFP43 modulates somatotrophic axis activity in female sheep
M. Szlis, B. J. Przybył, A. Wójcik-Gładysz
Journal of Animal and Feed Sciences
M. Szlis, B. J. Przybył, A. Wójcik-Gładysz
Journal of Animal and Feed Sciences
14.
The Mystery Actor in the Neuroendocrine Theater: Who Really Knows Obestatin? Central Focus on Hypothalamic–Pituitary Axes
Michał Szlis, Anna Wójcik-Gładysz, Alina Gajewska, Bartosz Jaroslaw Przybyl
International Journal of Molecular Sciences
Michał Szlis, Anna Wójcik-Gładysz, Alina Gajewska, Bartosz Jaroslaw Przybyl
International Journal of Molecular Sciences
15.
Tissue-Slice Organ-on-Chip Culture of Hypothalamic and Pituitary of Lambs─The Role of Phoenixin-20 as a Modulator of Gonadotrophic Axis
Michał Szlis, Bartosz Jarosław Przybył, Anna Wójcik-Gładysz
ACS Biomaterials Science & Engineering
Michał Szlis, Bartosz Jarosław Przybył, Anna Wójcik-Gładysz
ACS Biomaterials Science & Engineering
16.
CONSUMO DE BEBIDAS ENERGÉTICAS: UMA REVISÃO DOS RISCOS, EFEITOS ADVERSOS À SAÚDE
Ozanildo Vilaça do Nascimento, Milena Pantoja Aciole
Revista Multidisciplinar do Nordeste Mineiro
Ozanildo Vilaça do Nascimento, Milena Pantoja Aciole
Revista Multidisciplinar do Nordeste Mineiro
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.