Current issue
Online first
Archive
About the Journal
Editorial Office
Editorial Board
Copy right and self-archiving policy
Peer review process
Instructions for Reviewers
Printed version subscription
Abstracting and indexing
Contact
Instructions for Authors
Policies
General information
Open Access, Licensing terms, Commercial use and Copyright terms policies
Self-archiving policy and Archive policies
Article Correction and Withdrawal policy
Manuscript Submission policy
Authorship policy
Conflict of Interest policy
Language considerations policy
Plagiarism and Duplicate publications policy
Ethics policy
Review process policy
Acceptance of manuscripts policy
Online First Articles and Special Issues policies
Generative artificial intelligence (AI) policy
Advertising policy
Article publication charges
Policies
General information
Open Access, Licensing terms, Commercial use and Copyright terms policies
Self-archiving policy and Archive policies
Article Correction and Withdrawal policy
Manuscript Submission policy
Authorship policy
Conflict of Interest policy
Language considerations policy
Plagiarism and Duplicate publications policy
Ethics policy
Review process policy
Acceptance of manuscripts policy
Online First Articles and Special Issues policies
Generative artificial intelligence (AI) policy
Advertising policy
REVIEW PAPER
The effects of inulin supplementation in diets of pigs
on growth performance and selected meat quality traits:
a systematic review and meta-analysis
1
University of Siedlce, Institute of Animal Science and Fisheries, B. Prusa 14, 08-110 Siedlce, Poland
Publication date: 2026-05-06
Corresponding author
A. Milczarek
University of Siedlce, Institute of Animal Science and Fisheries, B. Prusa 14, 08-110 Siedlce, Poland
University of Siedlce, Institute of Animal Science and Fisheries, B. Prusa 14, 08-110 Siedlce, Poland
J. Anim. Feed Sci. 2026;35(2):e26
KEYWORDS
TOPICS
ABSTRACT
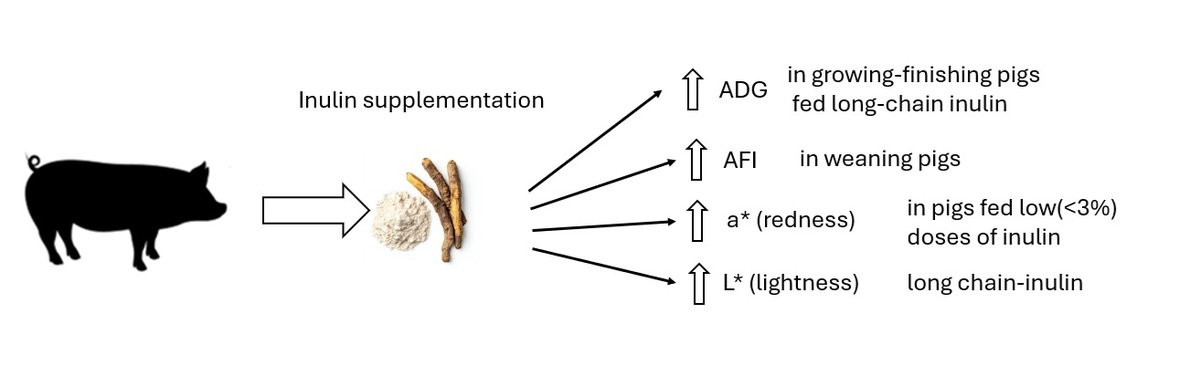
Inulin supplementation has been reported to enhance performance
responses in pigs and positively modify the quality of pork. However, the results
of research are inconsistent. The present study aimed to quantify the effects
of inulin supplementation in diets of pigs on performance traits and selected
pork quality traits, using the meta-analytical approach. Data were extracted from
45 studies and analysed using the random-effect model to estimate the effects
of inulin on average daily gain (ADG), average daily feed intake (ADFI), feed
conversion ratio and selected pork quality traits including initial and ultimate
pH (pH1 and pHu), drip loss (DL) and colour. The meta-analysis detected the
influence of inulin on ADG and ADFI under several study factors. The inulin
supplementation significantly improved ADG in growing-finishing pigs but
the stronger effect was detected for the long-chain inulin. The dose of inulin
in diet (<3% and ≥3%) enhanced ADG in similar range of the effect. Inulin
supplementation decreased ADFI only in weaning pigs. There were no effects
of inulin on pH1, pHu and DL. However, inulin supplementation significantly
modified the colour of pork. A trend to increase in redness (a*) and decrease
in yellowness (b*) was detected in loins of pigs fed with lower (<3%) doses of
inulin, nonetheless the application of long-chain inulin may result in increase of
lightness (L*) of meat. In conclusion, the inulin supplementation may enhance
ADG and colour characteristics but depend on degree of polarisation and dose.
ACKNOWLEDGEMENTS
The manuscript was realized under the COOPERATION measure of the Rural Development Programme for 2014–2024 by the Agency for Restructuring and Modernisation of Agriculture (00119.DDD.6509.00063.2022.07).
CONFLICT OF INTEREST
The Authors declare that there is no conflict of interest.
REFERENCES (132)
1.
Aluwé M., Langendries K.C.M., Beakert K.M., Tuyttens F.A.M., De Brabander D.L., De Smet S., Millet S., 2013.Effect of surgical castration, immunocastration and chicory-diet on the meat quality and palatability of boars. Meat Sci. 94, 402–407, https://doi.org/10.1016/j.meat....
2.
Aluwé M., Heyrman E., Theis S., Sieland C., Thurman K., Millet S., 2017. Chicory fructans in pig diet reduce skatole in back fat of entire male pigs. Res. Vet. Sci. 115, 340–344, http://dx.doi.org/10.1016/j.rv....
3.
Apolinario A.C., de Lima Damasceno B.P.G., de Macedo Beltrao N.E., Pessoa A., Converti A., da Silva J.A., 2014. Inulin-type fructans: a review on different aspects of biochemical and pharmaceutical technology. Carbohyd. Polym. 101, 368–378, https://doi.org/10.1016/j.carb....
4.
Apple J.K., 2007. Effects of nutritional modifications on the water-holding capacity of fresh pork: A review. J. Anim. Breed. Genet. 124, 43–58 https://doi.org/10.1111/j.1439....
5.
Apple J.K., 2013. Swine nutrition and pork quality. In: Nutrition for Successful and Sustainable Swine Nutrition, Chiba L. (ed.). Wiley-Blackwell, Ames, Iowa, USA. 413–437, https://doi.org/10.1002/978111....
6.
Apple J.K., Roberts W.J., Maxwell C.V., Rakes L.K., Friesen K.G., Fakler T.M., 2007. Influence of dietary inclusion level of manganese on pork quality during retail display. Meat Sci. 75, 640–647, https://doi.org/10.1016/j.meat....
7.
Bakbergenuly I., Hoaglin D.C., Kulinskaya E., 2020. Estimation in meta-analyses of mean difference and standardized mean difference. Statistics in Medicine. 39, 171–191, https://doi.org/10.1002/sim.84....
8.
Barrett J.R., Innes G.K., Johnson K.A., Lhermie G., Ivanek R., Greiner Safi A., Lansing D. 2021. Consumer perceptions of antimicrobial use in animal husbandry: A scoping review. PLoS ONE. 16, e0261010, https://doi.org/10.1371/journa....
9.
Bergamaschi M., Maltecca C., Schillebeeckx C., McNulty N.P., Schwab C., Shull C., Fix J., Tiezzi F., 2020. Heritability and genome-wide association of swine gut microbiome features with growth and fatness parameters. Sci. Rep. 10, 10134, https://doi.org/10.1038/s41598....
10.
Birmani M.W., Nawab A., Ghani M.W., Li G., Xiao M., An L.A, 2019. Review: Role of inulin in animal nutrition. J. Food Technol. Res. 6, 18–27, https://doi.org/10.18488/journ....
11.
Borenstein M., Hedges L. V., Higgins J. P. T., Rothstein H. R., 2009. Introduction to meta‐analysis. Wiley, Chichester, UK, https://doi.org/10.1002/978047...
12.
Böhmer B.M., Branner G.R., Roth-Maier D.A., 2005. Precaecal and faecal digestibility of inulin (DP 10–12) or an inulin/Enterococcus faecium mix and effects on nutrient digestibility and microbial gut flora. J. Anim. Physiol. Anim. Nutr. (Berl.) 89, 388–396, https://doi.org/10.1111/j.1439....
13.
Bradford H., McKernan C., Elliott C., Dean M., 2021. Consumers’ perceptions and willingness to purchase pork labelled ‘raised without antibiotics’. Appetite 171, 105900, https://doi.org/10.1016/j.appe....
14.
Brambillasca S., Zunino P., Cajarville C., 2015. Addition of inulin, alfalfa and citruspulp in diets for piglets: Influence on nutritional and faecal parameters, intestinal organs, and colonic fermentation and bacterial populations. Livestock Sci. 178, 243–250, https://doi.org/10.1016/j.livs....
15.
Chen T.T., Chen D.W., Tian G., Zheng P., Mao X.B., Yu J., He J., Huang Z.Q., Luo Y.H., Luo J.Q., Yu B., 2020. Effects of soluble and insoluble dietary fiber supplementation on growth performance, nutrient digestibility, intestinal microbe and barrier function in weaning piglet. Anim. Feed Sci. Technol. 260, 114335, https://doi.org/10.1016/j.anif....
16.
Cho J.H., Lee S.I., Kim I.H., 2015. Effects of different levels of fibre and benzoic acid on growth performance, nutrient digestibility, reduction of noxious gases, serum metabolites and meat quality in finishing pigs, J. Appl. Anim. Res. 43, 336–344, https://doi.org/10.1080/097121....
17.
Choe J. H., Choi Y. M., Lee S. H., Shin,H. G., Ryu Y. C., Hong K. C., Kim B. C., 2008. The relationship between glycogen, lactate content and muscle fiber type composition, and their influence on postmortem glycolytic rate and pork quality. Meat Sci. 80, 355–362, https://doi.org/10.1016/j.meat....
18.
Deng Y., Liu Y., Zhang Y., Wang Y., Liang J.B., Tufarelli V., Laudadio V., Liao X., 2017. Efficacy and role of inulin in mitigation of enteric sulfur-containing odor in pigs. J. Sci. Food Agric. 97, 2382–2391, https://doi.org/10.1002/jsfa.8....
19.
Denver S., Jensen J.D., Christensen T., 2021. Consumer preferences for reduced antibiotic use in Danish pig production. Prev. Vet. Med. 189, 105310, https://doi.org/10.1016/j.prev....
20.
Dunshea F.R., Pluske J.R., Ponnampalam E.N., 2024. Dietary iron or inulin supplementation alters iron status, growth performance, intramuscular fat and meat quality in finisher pigs. Meat Sci. 213, 109496, https://doi.org/10.1016/j.meat....
21.
Eberhard M., Hennig U., Kuhla S., Brunner R.M., Kleessen B., Metges C.C., 2007. Effect of inulin supplementation on selected gastric, duodenal, and caecal microbiota and short chain fatty acid pattern in growing piglets. Arch. Anim. Nutr. 61, 235–246, https://doi.org/10.1080/174503....
22.
Flickinger E.A., Van Loo J., Fahey G.C. Jr., 2003. Nutritional responses to the presence of inulin and oligofructose in the diets of domesticated animals: A review. Crit. Rev. Food Sci. Nutr. 43, 19–60, https://doi.org/10.1080/104086....
23.
Gagaoua M., Suman S. P., Purslow P. P., Lebret B., 2023. The color of fresh pork: Consumers expectations, underlying farm-to-fork factors, myoglobin chemistry and contribution of proteomics to decipher the biochemical mechanisms. Meat Sci. 206, 109340, https://doi.org/10.1016/j.meat....
24.
Gardiner G.E., Metzler-Zebeli B.U., Lawlor P.G., 2020. Impact of intestinal microbiota on growth and feed efficiency in pigs: a review. Microorganisms 8, 1886, https://doi.org/10.3390/microo....
25.
Gibson G.R., Scott K.P., Rastall R.A. et al., 2010. Dietary prebiotics: Current status and new definition. Food Sci. Technol. Bull. Funct. Foods 7, 1–19, https://doi.org/10.1616/1476-2....
26.
Grela E.R., Pietrzak K., Sobolewska S., Witkowski P., 2013. Effect of inulin and garlic supplementation in pig diets. Ann Anim Sci. 13, 63–71, https://doi.org/10.2478/v10220....
27.
Grela E.R., Sobolewska S., Kowalczuk-Vasilev E., Krasucki W., 2014. Effect of dietary inulin source on piglet performance, immunoglobulin concentration, and plasma lipid profile. Bull Vet Inst Pulawy 58, 453–458, https://doi.org/10.2478/bvip-2....
28.
Grela E.R., Swiatkiewicz M., Florek M., Bakowski M., Skiba G., 2021. Effect of inulin source and a probiotic supplement in pig diets on carcass traits, meat quality and fatty acid composition in finishing pigs. Animals 11, 2438, https://doi.org/10.3390/ani110....
29.
Grunert K.G., Bredahl L., Brunsø K., 2004. Consumer perception of meat quality and implications for product development in the meat sector – A review. Meat Sci. 66, 259–272, https://doi.org/10.1016/S0309-....
30.
Grunert K.G., Sonntag W.I., Glanz-Chanos V., Forum S., 2018. Consumer interest in environmental impact, safety, health and animal welfare aspects of modern pig production: Results of a cross-national choice experiment. Meat Sci. 137, 123–129, https://doi.org/10.1016/j.meat....
31.
Halas D., Hansen C.F., Hampson D.J., Mullan B.P., Wilson R.H., Pluske J.R. 2009. Effect of dietary supplementation with inulin and/or benzoic acid on the incidence and severity of post-weaning diarrhoea in weaner pigs after experimental challenge with enterotoxigenic Escherichia coli. Arch. Anim. Nutr., 63, 267–280, https://doi.org/10.1080/174503....
32.
Han Q., Huang X., Yan F., Yin J., Xiao Y., 2022. The role of gut microbiota in the skeletal muscle development and fat deposition in pigs. Antibiotics. 11, 793, https://doi.org/10.3390/antibi....
33.
Han P., Li P., Zhou W., Fan L., Wang B., Liu H., Gao C., Du T., Pu G., Wu C., Zhang Z., Niu P., Huang R., Li H., 2020. Effects of various levels of dietary fiber on carcass traits, meat quality and myosin heavy chain I, IIa, IIx and IIb expression in muscles in erhualian and large white pigs. Meat Sci. 169, 108160, https://doi.org/10.1016/j.meat....
34.
Hansen L. L., Agerhem H., Rosenvold K., Jensen M. T., 2002. Effect of Brussels sprouts and inulin/rape seed cake on the sensory profile of pork M. longissimus dorsi. Meat Sci. 61, 441–448, https://doi.org/10.1016/S0309-....
35.
Hansen L.L., Stolzenbach S., Jensen J.A., Henckel P., Hansen-Moller J., Syriopoulos K., Byrne D.V., 2008. Effect of feeding fermentable fibre-rich feedstuffs on meat quality with emphasis on chemical and sensory boar taint in entire male and female pigs. Meat Sci. 80, 1165–1173, https://doi.org/10.1016/j.meat....
36.
He J., Xie H., Chen D., Yu B., Huang Z., Mao X., Zheng P., Luo Y., Yu J., Luo J., Yan H., 2021. Synergetic responses of intestinal microbiota and epithelium to dietary inulin supplementation in pigs. Eur. J. Nutr. 60, 715–727, https://doi.org/10.1007/s00394....
37.
Henchion M., McCarthy M., Resconi V. C., Troy D., 2014. Meat consumption: Trends and quality matters. Meat Sci. 98, 561–568, https://doi.org/10.1016/j.meat....
38.
Herosimczyk A., Lepczyński A., Werkowska M. et al., 2022. Dietary inclusion of dried chicory root affects cecal mucosa proteome of nursery pigs. Animals 12, 1710, https://doi.org/10.3390/ani121....
39.
Higgins J. P. T., Thompson S. G., 2002. Quantifying heterogeneity in a meta‐analysis. Stat. Med. 21, 1539–1558, https://doi.org/10.1002/sim.11....
40.
Higgins J.P., Thompson S.G., Spiegelhalter D.J., 2009. A re-evaluation of random-effects meta-analysis. J. R. Stat. Soc. Ser. A Stat. Soc. 172, 137–159, https://doi.org/10.1111/j.1467....
41.
Hu Y., Chen D., Yu B., Yan H., Zheng P., Mao X., Yu J., He J., Huang Z., Luo Y., Luo J., Zhang X., Luo L., 2020. Effects of dietary fibres on gut microbial metabolites and liver lipid metabolism in growing pigs. J. Anim. Physiol. Anim. Nutr. 104, 1484–1493, https://doi.org/10.1111/jpn.13....
42.
Hughes J.M., Oiseth S.K., Purslow P.P., Warner R.D., 2014. A structural approach to understanding the interactions between colour, water-holding capacity and tenderness. Meat Sci. 98, 520–532, https://doi.org/10.1016/j.meat....
43.
Ivarsson E., Liu H.Y., Dicksved J., Roos S., Lindberg J.E., 2012. Impact of chicory inclusion in a cereal-based diet on digestibility, organ size and faecal microbiota in growing pigs. Anim. Int. J. Anim. Biosci. 6, 1077–1085, https://doi.org/10.1017/S17517....
44.
Jayasooriya S.D., Pluske J.R., Dunshea F.R., Gill H., Ponnampalam E.N., 2009. Dietary iron improves iron status in finisher pigs fed wheat based diets. Manipulating Pig Production XII Proceedings of the Twelfth Biennial Conference of the Australasian Pig Science Association (APSA). Publisher Australasian Pig Science Association, ISBN 978-0-9806880-0-9.
45.
Jolliff J., Mahan D., 2012. Effect of dietary inulin and phytase on mineral digestibility and tissue retention in weanling and growing swine. Anim. Sci. 90, 3012–3022, https://doi.org/10.2527/jas.20....
46.
Joo S.T., Kim G.D., Hwang Y.H., Ryu Y.C., 2013. Control of fresh meat quality through manipulation of muscle fiber characteristics. Meat Sci. 95, 828–836, https://doi.org/10.1016/j.meat....
47.
Kozłowska I., Marć-Pieńkowska J., Bednarczyk M., 2016. Beneficial aspects of inulin supplementation as a fructooligosaccharide prebiotic in monogastric animal nutrition—a review. Ann Anim Sci. 16, 315–331, https://doi.org/doi.org/10.151....
48.
Lan Q., Lian Y., Peng P., Yang L., Zhao H., Huang P., Ma H., Wei H., Yin Y., Liu M., 2023. Association of gut microbiota and SCFAs with finishing weight of Diannan small ear pigs. Front. Microbiol. 14, 1117965, https://doi.org/10.3389/fmicb.....
49.
Lee J.H., Kim S., Kim E.S., Keum G.B., Doo H., Kwak J., Pandey S., Cho J.H., Ryu S., Song M., Cho J.H., Kim S., Kim H.B., 2023. Comparative analysis of the pig gut microbiome associated with the pig growth performance. J. Anim. Sci. Technol. 65, 856–864, https://doi.org/10.5187/jast.2....
50.
Lefaucheur L., 2010. A second look into fibre typing — Relation to meat quality. Meat Sci. 84, 257–270, https://doi.org/10.1016/j.meat....
51.
Lefaucheur L., Ecolan P., Plantard L., Gueguen N., 2002. New insights into muscle fiber types in the pig. J. Histochem. Cytochem. 50, 719–730, https://doi.org/10.1177/002215....
52.
Lepczyński A., Herosimczyk A., Barszcz M., Ożgo M., Michałek K., Grabowska M., Tuśnio A., Szczerbińska D., Skomiał J., 2021. Diet supplemented either with dried chicory root or chicory inulin significantly influence kidney and liver mineral content and antioxidative capacity in growing pigs. Animal 15, 100129, https://doi.org/10.1016/j.anim....
53.
Li Y., Li J., Zhang L., Yu C., Lin M., Gao F., Zhou G., Zhang Y., Fan Y., Nuldnali L., 2015. Effects of dietary energy sources on post mortem glycolysis, meat quality and muscle fibre type transformation of finishing pigs. PLoS ONE 10, e0131958, https://doi.org/10.1371/journa....
54.
Li H., Yin J., Tan B., Chen J., Zhang H., Li Z., Ma X., 2021. Physiological function and application of dietary fiber in pig nutrition: A review. Anim. Nutr. 7, 259–267, https://doi.org/10.1016/j.anin....
55.
Lin-Schilstra L., Backus G., Snoek H., Mörlein D., 2022. Consumers’ view on pork: consumption motives and production preferences in ten European Union and four non-European Union countries. Meat Sci. 187, 108736, https://doi.org/10.1016/j.meat....
56.
Liu B., Han R.H. Hao H., Wang Z., Yong F., Li H., Zhang T., Yang L., Jiang H., Che D., 2024. Interaction of dietary fiber and soluble to insoluble dietary fiber ratio in improving production performance, meat quality and muscle fiber type transformation in finishing pigs, https://doi.org/10.2139/ssrn.4....
57.
Liu Y., Espinosa C.D., Abelilla J.J., Casas G.A., Lagos L.V., Lee S.A., Kwon W.B., Mathai J.K., Navarro D.M.D.L., Jaworski N.W., Stein H.H., 2018. Non-antibiotic feed additives in diets for pigs: a review. Anim Nutr. 4, 113–25, https://doi.org/10.1016/j.anin....
58.
Liu N., Shen H., Zhang F., Liu X., Xiao Q., Jiang Q., Tan B., Ma X., 2023. Applications and prospects of functional oligosaccharides in pig nutrition: a review. Anim Nutr. 13, 206–215, https://doi.org/10.1016/j.anin....
59.
Loh G., Eberhard M., Brunner R.M., Hennig U., Kuhla S., Kleessen B., Metges C.C., 2006. Inulin alters the intestinal microbiota and short-chain fatty acid concentrations in growing pigs regardless of their basal diet. J. Nutr. 136, 1198–1202, https://doi.org/10.1093/jn/136....
60.
Luo R., Song X., Li Z., Zhang A., Yan X., Pang Q., 2018. Effect of soil salinity on fructan content and polymerization degree in the sprouting tubers of Jerusalem artichoke (Helianthus tuberosus L.). Plant Physiol. Biochem. 125, 27–34, https://doi.org/10.1016/j.bcdf....
61.
Lynch M.B., Sweeney T., Callan B.F.J.J., O’Doherty J.V., 2007. The effect of high and low dietary crude protein and inulin supplementation on nutrient digestibility, nitrogen excretion, intestinal microflora and manure ammonia emissions from finisher pigs. Animal 1, 1112–1121, https://doi.org/10.1017/S17517....
62.
Ma J., Piao X., Mahfuz S., Long S., Wang J., 2022. The interaction among gut microbes, the intestinal barrier and short chain fatty acids Anim. Nutr. 9, 159–174, https://doi.org/10.1016/j.anin....
63.
MacFabe D.F., Cain D.P., Rodriguez-Capote K., Franklin A.E., Hoffman J.E., Boon F., Taylor A.R., Kavaliers M., Ossenkopp K.P., 2007. Neurobiological effects of intraventricular propionic acid in rats: possible role of short chain fatty acids on the pathogenesis and characteristics of autism spectrum disorders. Behav. Brain Res. 176, 149–169, https://doi:10.1016/j.bbr.2006....
64.
Mach N., Berri M., Estellé J., Levenez F., Lemonnier G., Denis C., Leplat J.J., Chevaleyre C., Billon Y., Doré J., Rogel-Gaillard C., Lepage P., 2015. Early-life establishment of the swine gut microbiome and impact on host phenotypes. Environ Microbiol Rep. 7, 554–569, https://doi:10.1111/1758-2229.....
65.
Mair C., Plitzner C., Domig K. J., Schedle K., Windisch W., 2010. Impact of inulin and a multispecies probiotic formulation on performance, microbial ecology and concomitant fermentation patterns in newly weaned piglets. J. Anim. Physiol. Anim. Nutr. 94, 164–177e, https://doi:10.1111/j.1439-039....
66.
Mancini R.A., Hunt M.C., 2005. Current research in meat color. Meat Sci. 71, 100–121, https://doi.org/10.1016/j.meat....
67.
McCormack U.M., Curiăo T., Metzler-Zebeli B.U., Wilkinson T., Reyer H., Crispie F., Cotter P.D., Creevey C.J., Gardiner G.E., Lawlor P.G., 2019. Improvement of feed efficiency in pigs through microbial modulation via fecal microbiota transplantation in sows and dietary supplementation of inulin in offspring. Appl. Environ. Microbiol. 85, e01255-19, https://doi.org/10.1128/AEM.01....
68.
Meerza S.I.A., Gulab S., Brooks K.R., Gustafson C.R., Yiannaka A. 2022. U.S. consumer attitudes toward antibiotic use in livestock production. Sustainability. 14, 7035, https://doi.org/10.3390/su1412....
69.
Metzler-Zebeli B.U., Trevisi P., Prates J.A.M., Tanghe S., Bosi P., Canibe N., Montagne L., Freir J., Zebeli Q., 2017. Assessing the effect of dietary inulin supplementation on gastrointestinal fermentation, digestibility and growth in pigs: A meta-analysis. Anim. Feed Sci. Technol. 233, 120–132, http://dx.doi.org/10.1016/j.an....
70.
Miao M., Dai Y., Ru C., Fan Y., Wang X., Fan C., Mu J., Hou W., Dong Z., Li P., Sun G., Zeng X., 2021. Dietary supplementation of inulin alleviates metabolism disorders in gestational diabetes mellitus mice via RENT/AKT/IRS/GLUT4 pathway. Diabetol. Metab. Syndr. 13, 150, https://doi.org/10.1186/s13098....
71.
Mikkelsen L.L., Jakobsen M., Jensen, B.B., 2003. Effects of dietary oligosaccharides on microbial diversity and fructo-oligosaccharide degrading bacteria in faeces of piglets post-weaning. Anim. Feed. Sci. Technol. 109, 133–150, https://doi.org/10.1016/S0377-....
72.
Niness K.R., 1999. Inulin and oligofructose: what are they? J. Nutr. 129, 1402–1406, https://doi.org/10.1093/jn/129....
73.
Nowak P., Kasprowicz-Potocka M., Zaworska A. et al., 2017. The effect of eubiotic feed additives on the performance of growing pigs and the activity of intestinal microflora. Arch. Anim. Nutr. 71, 455–469, https://doi.org/10.1080/174503....
74.
OECD/Food and Agriculture Organization of the United Nations 2023. “Meat”, in OECD-FAO Agricultural Outlook 2023-2032, OECD Publishing, Paris. https://doi.org/10.1787/08801a....
75.
Oh J. K., Chae J. P., Pajarillo E. A. B., Kim S. H., Kwak M. J., Eun J. S., Chee S.W., Whang K.Y., Kim S.H., Kang D.K., 2020. Association between the body weight of growing pigs and the functional capacity of their gut microbiota. Anim. Sci. J. 91, e13418, https://doi.org/10.1111/asj.13....
76.
Ojima M. N., Yoshida K., Sakanaka M., Jiang L., Odamaki T., Katayama T., 2022. Ecological and molecular perspectives on responders and non-responders to probiotics and prebiotics. Curr. Opin. Biotechnol. 73, 108–120, https://doi.org/10.1016/j.copb....
77.
Øverland M., Kjos, N.K., Fauske A.K., Teige J., Sørum H., 2011. Easily fermentable carbohydrates reduce skatole formation in the distal intestine of entire male pigs. Livest. Sci. 40, 206–217, https://doi.org/10.1016/j.livs....
78.
Patience J.F., Ramirez A., 2022. Invited review: strategic adoption of antibiotic-free pork production: the importance of a holistic approach. Transl. Anim. Sci. 6, txac063, https://doi.org/10.1093/tas/tx....
79.
Patterson J.K., Yasuda K., Welch R.M., Miller D.D., Lei X.G., 2010. Supplemental dietary inulin of variable chain lengths alters intestinal bacterial populations in young pigs. J. Nutr. 140, 2158–2161, https://doi.org/10.3945/jn.110....
80.
Paudel B., Kolady D., Grebitus C., Roy A., Ishaq M., 2022. Consumers’ willingness to pay for pork produced with different levels of antibiotics. Q Open 2, qoac001, https://doi.org/10.1093/qopen/....
81.
Pierce K.M., Callan J.J., McCarthy P., O’Doherty J.V., 2005. Performance of weanling pigs offered low or high lactose diets supplemented with avilamycin or inulin. Anim. Sci. 80, 313–318, https://doi.org/10.1079/ASC409....
82.
Ponnampalam E., Jayasooriya D., Gill H., Pluske J.R., Dunshea F.R., 2009. Nutritional manipulation of iron level in finisher pigs and fresh pork. Final Report (3A-108) prepared for the Cooperative Research Centre for an Internationally Competitive Pork Industry.
83.
Przybylski W., Jaworska D., Sałek P., Sobol M., Branicki M., Skiba G., Raj S., Jankiewicz U., 2019. The effect of inulin supply to high‐fat diet rich in saturated fatty acids on pork quality and profile of sarcoplasmic protein in meat exudate. J. Anim. Physiol. Anim. Nutr. 103, 593–602, https://doi.org/10.1111/jpn.13....
84.
Purslow P.P., Gagaoua M., Warner R. D., 2021. Insights on meat quality from combining traditional studies and proteomics. Meat Sci., 174, 108423, https://doi.org/10.1016/j.meat....
85.
Qin Y.Q., Wang L.Y., Yang X.Y., Xu Y.J., Fan G., Fan Y.G., Ren J.N., Li Q.X., 2023. Inulin: properties and health benefits. Food Funct. 14, 2948, https://doi.org/10.1039/d2fo01....
86.
Richardson M., Garner P., Donegan S., 2019. Interpretation of subgroup analyses in systematic reviews: A tutorial. Clin. Epidemiol. Glob. Health, 7, 2, 192–198, https://doi.org/10.1016/j.cegh....
87.
Roberfroid M.B., Vanloo J.A.E., Gibson G.R., 1998. The bifidogenic nature of chicory inulin and its hydrolysis products. J. Nutr. 128, 11–19, https://doi.org/10.1093/jn/128....
88.
Roberfroid M. B., 2005. Introducing inulin-type fructans. Br. J. Nutr. 93, (Suppl 1), 13–25, https://doi.org/10.1079/bjn200....
89.
Roberfroid M. B., 2007. Inulin-type fructans: functional food ingredients. J. Nutr. 137, 2493–2502, https://doi.org/10.1093/jn/137....
90.
Rosenvold K., Andersen H.J., 2003. The significance of pre-slaughter stress and diet on colour and colour stability of pork. Meat Sci. 63, 199–209, https://doi.org/10.1016/S0309-....
91.
Rosenvold K., Essen‐Gustavsson B., Andersen H. J., 2003. Dietary manipulation of pro‐ and macroglycogen in porcine skeletal muscle. J. Anim. Sci. 81, 130–134, https://doi.org/10.2527/2003.8....
92.
Rosenvold K., Lærke H.N., Jensen S.K.; Karlsson A.H., Lundström K., Andersen H.J., 2001a. Strategic finishing feeding as a tool in the control of pork quality. Meat Sci. 59, 397–406, https://doi.org/10.1016/S0309-....
93.
Rosenvold K., Laerke H. N., Jensen S. K., Karlsson A. H., Lundstrom K., Andersen H. J., 2002. Manipulation of critical quality indicators and attributes in pork through vitamin E supplementation, muscle glycogen reducing finishing feeding and preslaughter stress. Meat Sci. 62, 485–496, https://doi.org/10.1016/S0309-....
94.
Rosenvold K., Petersen J. S., Lærke H. N., Jensen S. K., Therkildsen M., Karlsson A. H., Møller H. S., Andersen H. J., 2001b. Muscle glycogen stores and meat quality as affected by strategic finishing feeding of slaughter pigs. J. Anim. Sci. 79, 382–391, https://doi.org/10.2527/2001.7....
95.
Samolińska W., Grela ER., 2017. Comparative effects of inulin with different polymerization degrees on growth performance, blood trace minerals, and erythrocyte indices in growing-finishing pigs. Biol. Trace Elem. Res. 176, 130–142, https://doi.org/10.1007/s12011....
96.
Samolińska W., Grela E.R., Kiczorowska B., 2019. Effects of inulin extracts and inulin-containing plants on haematobiochemical responses, plasma mineral concentrations, and carcass traits in growing-finishing pigs. J. Elem. 24, 711–726, https://doi.org/10.5601/jelem.....
97.
Samolińska W., Kowalczuk-Vasilev E., Grela E.R., 2018. Comparative effect of different dietary inulin sources and probiotics on growth performance and blood characteristics in growing-finishing pigs. Arch. Anim. Nutr. 72, 379–395, https://doi.org/10.1080/174503....
98.
San Andres J.V., Mastromano G.A., Li Y., Tran H., Bundy J.W., Miller P.S., Burkey T.E., 2019. The effects of prebiotics on growth performance and in vitro immune biomarkers in weaned pigs. Transl. Anim. Sci. 3, 1315–1325, https://doi.org/10.1093/tas/tx....
99.
Schafer A., Rosenvold K., Purslow P. P., Andersen H. J., Henckel P., 2002. Physiological and structural events post mortem of importance for drip loss in pork. Meat Sci. 61, 355–366, https://doi.org/10.1016/S0309-....
100.
Scheffler T. L., Gerrard D. E., 2007. Mechanisms controlling pork quality development: The biochemistry controlling postmortem energy metabolism. Meat Sci. 77, 7–16, https://doi.org/10.1016/j.meat....
101.
Shen Q. W., Means W. J., Thompson S. A., Underwood K. R., Zhu M. J., McCormick R. J., Ford S.P., Du M., 2006. Pre-slaughter transport, AMP activated protein kinase, glycolysis, and quality of pork loin. Meat Sci. 74, 388–395, https://doi.org/10.1016/j.meat....
102.
Shoaib M., Shehzad A., Omar M., Rakha A., Raza H., Sharif H. R., Shakeel A., Ansari A., Niazi S. 2016. Inulin: Properties, health benefits and food applications. Carbohydr. Polym. 147, 444–454, https://doi.org/10.1016/j.carb....
103.
Sobol M., Raj S., Skiba G., 2018. Inulin supplementation reduces the negative effect of a high‐fat diet rich in SFA on bone health of growing pigs. Br. J. Nutr. 119, 1111–1118, https://doi.org/10.1017/S00071....
104.
Sobolewska S., Grela E.R., 2014. The effect of inulin extraction method or powder from inulin-producing plants in fattener diets on performance, carcass traits and meat quality. Ann. Anim. Sci. 14, 911–920, https://doi.org/10.2478/aoas-2....
105.
Sobolewska S., Grela E.R., 2015. Effect of inulin extract or inulin-containing plant supplement on short-chain fatty acids in the large intestine and jejunum histology of fatteners. Med. Wet. 71, 432–435.
106.
Stewart M.L., Timm D.A., Slavin J.L., 2008. Fructooligosaccharides exhibit more rapid fermentation than long-chain inulin in an in vitro fermentation system. Nutr Res. 28, 329–334, https://doi.org/10.1016/j.nutr....
107.
Tang S., Xin Y., Ma Y., Xu X., Zhao S., Cao J., 2020. Screening of microbes associated with swine growth and fat deposition traits across the intestinal tract. Front Microbiol. 11, 586776, https://doi.org/10.3389/fmicb.....
108.
Tako E., Glahn R.P., Welch R.M. Lei X., Yasuda K., Miller D.D., 2008. Dietary inulin affects the expression of intestinal enterocyte iron transporters, receptors and storage protein and alters the microbiota in the pig intestine. Br. J. Nutr. 99, 472–480, https://doi.org/10.1017/S00071....
109.
Topping D.L., Clifton P.M., 2001. Short-chain fatty acids and human colonic function: roles of resistant starch and nonstarch polysaccharides. Physiol Rev. 81, 1031–1064, https://doi.org/10.1152/physre....
110.
Trefan, L., Bünger, L., Rooke, J., Blom-Hansen, J., Salmi, B., Larzul, C., Terlouw, C., and Doeschl-Wilson, A. 2010. Meta-analysis of effects of dietary vitamin E and post slaughter storage conditions on changes of redness (a*) of pork, Arch. Anim. Breed. 53, 564–577, https://doi.org/10.5194/aab-53....
111.
Varley P.F., McCarney C., Callan J.J., O'Doherty J.V., 2010. Effect of dietary mineral level and inulin inclusion on phosphorus, calcium and nitrogen utilisation, intestinal microflora and bone development. J. Sci. Food Agric. 90, 2447–2454, https://doi.org/10.1002/jsfa.4....
112.
Vesterinen H., Sena E., Egan K., Hirst T., Churolov L., Currie G., Antonic A., Howells D., MacLeod M., 2014. Meta-analysis of data from animal studies: A practical guide. J. Neurosci. Methods 221, 92–102, https://doi.org/10.1016/j.jneu....
113.
Vhile S.G., Kjos N.P., Sørum H., Øverland M., 2012. Feeding Jerusalem artichoke reduced skatole level and changed intestinal microbiota in the gut of entire male pigs. Animal. 6, 807–814, https://doi.org/10.1017/S17517....
114.
Wang W., Chen D., Yu B., Huang Z., Luo Y., Zheng P., Mao X., Yu J., Luo J., He J., 2019. Effect of dietary inulin supplementation on growth performance, carcass traits, and meat quality in growing–finishing pigs. Animals. 9, 840, https://doi.org/10.3390/ani910....
115.
Wang W., Chen D., Yu B., Huang Z., Mao X., Zheng P., Luo Y., Yu J., Luo J., Yan H., He J., 2020. Effects of dietary inulin supplementation on growth performance, intestinal barrier integrity and microbial populations in weaned pigs. Br. J. Nutr. 124, 296–305, https://doi.org/10.1017/S00071....
116.
Wang L., Huang Y., Wang Y., Shan T., 2021. Effects of polyunsaturated fatty acids supplementation on the meat quality of pigs: A meta-analysis. Front. Nutr. 8, 746765, https://doi.org/10.3389/fnut.2....
117.
Warner R.D., Greenwood P. L., Pethick D.W., Ferguson D.M., 2010. Genetic and environmental effects on meat quality. Meat Sci. 86, 171–183, https://doi.org/10.1016/j.meat....
118.
Wellock I.J., Fortomaris P.D., Houdijk J.G.M., Wiseman J., Kyriazakis I., 2008. The consequences of non-starch polysaccharide solubility and inclusion level on the health and performance of weaned pigs challenged with enterotoxigenic Escherichia coli. Br. J. Nutr. 99, 520–530, https://doi.org/10.1017/S00071....
119.
Wen C., Wang Q., Gu S., Jin J., Yang N., 2024. Emerging perspectives in the gut–muscle axis: The gut microbiota and its metabolites as important modulators of meat quality. Microb. Biotechnol. 17, e14361. https://doi.org/10.1111/1751-7....
120.
Wilborn B. S., Kerth C. R., Owsley W. F., Jones W. R., Frobish L. T., 2004. Improving pork quality by feeding supranutritional concentrations of vitamin D31. J. Anim. Sci. 82, 218–224, https://doi.org/10.2527/2004.8....
121.
Wu W., Zhang L., Xia B., Tang S., Liu L., Xie J., Zhang H., 2020. Bioregional alterations in gut microbiome contribute to the plasma metabolomic changes in pigs fed with inulin. Microorganisms 8, 111, https://doi.org/10.3390/microo....
122.
Xia B., Wu W., Zhang L., Wen X., Xie J., Zhang H., 2021. Gut microbiota mediates the effects of inulin on enhancing sulfomucin production and mucosal barrier function in a pig model. Food Funct. 12, 10967–10982, https://doi.org/10.1039/d1fo02....
123.
Yan E., Guo J., Yin J., 2023. Nutritional regulation of skeletal muscle energy metabolism, lipid accumulation and meat quality in pigs. Adv. Nutr. 14, 185–192, https://doi.org/10.1016/j.anin....
124.
Yang Y., Liu Y., Liu J., Wang H., Guo Y., Du M., Cai C., Zhao Y., Lu C., Guo X., Cao G., Duan Z., Li B., Gao p., 2021. Composition of the fecal microbiota of piglets at various growth stages. Front. Vet. Sci. 8, 661671, https://doi.org/10.3389/fvets.....
125.
Yang C., Lu H., Li E., Oladele P., Ajuwon K.M., 2023. Inulin supplementation induces expression of hypothalamic antioxidant defence genes in weaned piglets. J. Anim. Physiol. Anim. Nutr. 107, 157–164, https://doi.org/10.1111/jpn.13....
126.
Yasuda K., Dawson H.D., Wasmuth E.V., Roneker C., Chen C., Urban J.F., Welch R.M., Miller D.D., Lei X.G., 2009. Supplemental dietary inulin influences expression of iron and inflammation related genes in young pigs. J. Nutr. 139, 2018–2023, https://doi.org/10.3945/jn.109....
127.
Yasuda K., Maiorano R., Welch R. M., Miller D. D., Lei X. G., 2007. Cecum is the major degradation site of ingested inulin in young pigs. J. Nutr. 137, 2399–2404, https://doi.org/10.1093/jn/137....
128.
Yasuda K., Roneker K.R., Miller D.D., Welch R.M., Lei X.G., 2006. Supplemental dietary inulin affects the bioavailability of iron in corn and soybean meal to young pigs. J. Nutr. 136, 3033–3038, https://doi.org/10.1093/jn/136....
129.
Yin P., Du T., Yi S., Zhang Ch., Yu L., Tian F., Chen W., Zhai Q., 2023. Response differences of gut microbiota in oligofructose and inulin are determined by the initial gut Bacteroides/Bifidobacterium ratios. Food Res. Int. 174, 1, 113598, https://doi.org/10.1016/j.food....
130.
Zhang Y., Yu B., Yu J., Zheng P., Huang Z., Luo Y., Luo J., Mao X., Yan H., He J., Chen D., 2019. Butyrate promotes slow-twitch myofiber formation and mitochondrial biogenesis in finishing pigs via inducing specific microRNAs and PGC-1α expression1. J Anim Sci. 97, 3180–3192, https://doi.org/10.1093/jas/sk....
131.
Zhou H., Yu B., Sun J., Liu Z., Chen H., Ge L., Chen D., 2021. Short-chain fatty acids can improve lipid and glucose metabolism independently of the pig gut microbiota. J. Anim. Sci. Biotechnol. 12, 61, https://doi.org/10.1186/s40104....
132.
Zlowodzki M., Poolman R. W., Kerkhoffs G. M., Tornetta P., Bhandari M., 2007. How to interpret a meta-analysis and judge its value as a guide for clinical practice. Acta Orthop. 78, 598–609, https://doi.org/10.1080/17453 670710014284.
Share
RELATED ARTICLE
We process personal data collected when visiting the website. The function of obtaining information about users and their behavior is carried out by voluntarily entered information in forms and saving cookies in end devices. Data, including cookies, are used to provide services, improve the user experience and to analyze the traffic in accordance with the Privacy policy. Data are also collected and processed by Google Analytics tool (more).
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.
You can change cookies settings in your browser. Restricted use of cookies in the browser configuration may affect some functionalities of the website.